Description
Pregnacare® Plus combines the original Pregnacare® tablet formula, the leading supplement in pregnancy care, with high quality Omega-3 (DHA 300mg) capsules, in a convenient 2-in-1 pack, for use throughout pregnancy.
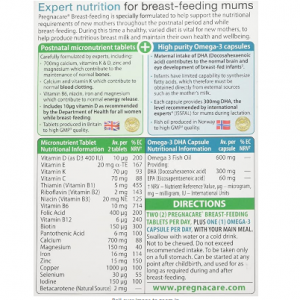
Each Pregnacare® tablet provides a wide range of essential vitamins and minerals including the exact recommended levels of 400µg folic acid and 10µg vitamin D. Supplemental folic acid 2 intake increases maternal folate status. Low maternal folate status is a risk factor in the development of neural tube defects in the developing foetus. Moderate iron and vitamins B6 and B12 which contribute to normal red blood cell formation. Nutritional tablets produced in Britain to high GMP 3 quality.
Each high purity Omega-3 capsule provides 300mg DHA (Docosahexaenoic Acid), a level recommended by many international experts 4 (ISSFAL). Maternal intake of DHA contributes to the normal development of the eye & brain of the foetus. Fish oil produced to high GMP 3 quality. Pregnacare® does not contain vitamin A, as high levels are not advised in pregnancy.
Dosage: One tablet (green blister pack) plus one capsule (pink blister pack) per day, with a main meal. There is no need for an additional multivitamin.
Composition: 28 Tablets containing essential minerals and vitamins and 400µg folic acid and 10µg vitamin D and 28 Omega-3 Capsules.
Side Effects: Diarrhea. Upset stomach. Abdominal cramping. Decreased appetite. Changes in stool color or texture.













Reviews
There are no reviews yet.